The value of reaction quotient (Q) , for the following cell, Zn(s) | Zn^2 + (0.01 M)|| Ag^+ (1.25 M)|Ag(s) is:

How to calculate $q_{1}(t)$, $q_{2}(t)$, $p_{1}(t)$, $p_{2}(t)$ using Euler's method? - Mathematics Stack Exchange

Michael 🧪 Farabaugh on Twitter: "@louiebarnett123 (3/5) Let's see what happens when the volume of the reaction vessel is doubled (at constant T). When we calculate Q, we see that Q <
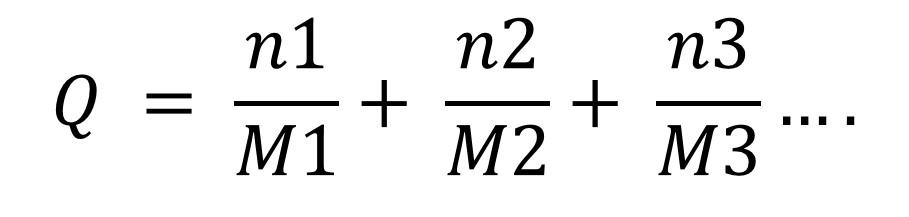
Calculating the "Q" Value for Different Dangerous Goods in a Single Outer Packaging (All Packed in One) - Daniels Training Services